Direct Current Fundamentals
Introduction to Electricity And Electronics
-
Question 1
How do we define the terms positive charge and negative charge?
Reveal answerIt is now known that the process of charging is the transfer of electrons. Thus, a positively charged object has lost some of its normal number of electrons. And a negatively charged object has been given extra electrons so that it has more than its normal number of electrons.
-
Question 2
Using what you know of electron theory, explain what must happen to give an object a positive charge. What happens to give an object a negative charge?
Reveal answerAny ordinary object contains an equal number of electrons (negative charge) and protons (positive charge). If some of the electrons are removed, the object is called positively charged. If electrons are added to the neutral object, it becomes negatively charged.
-
Question 3
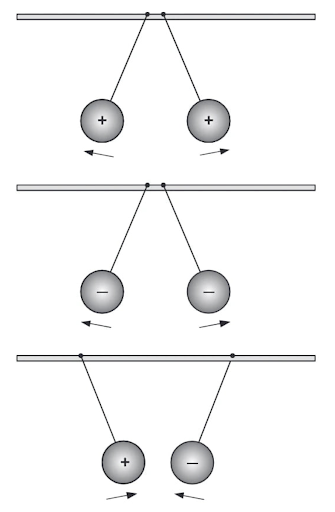
State the law of attraction and repulsion.
Reveal answerThe Law of Attraction and Repulsion states that like charges repel and unlike charges attract each other.
-
Question 4
What kind of charge does an electron have?
Reveal answerElectrons have a negative charge.
-
Question 5
Would two electrons repel or attract each other? Explain.
Reveal answerElectrons will repel each other because electrons are all negatively charged, and like charges repel each other.
-
Question 6
What do each of these words mean: atom, element, molecule?
Reveal answerAn atom is the smallest part of an element. Atoms are made of electrons, protons, and neutrons.(One exception to this description of atoms is the atom of ordinary hydrogen which does not have neutrons.)
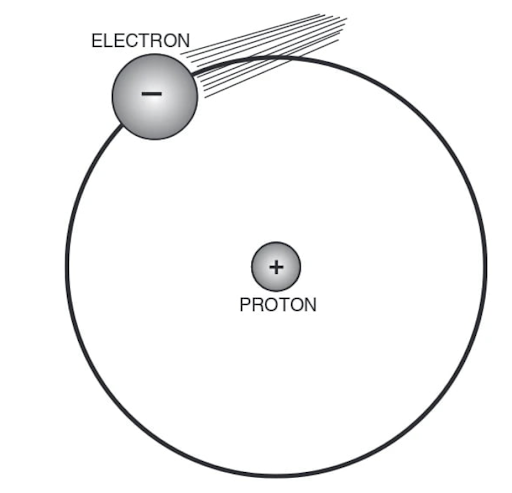
Hydrogen Atom
Elements are materials that consist of only one kind of atom. Elements cannot be broken down into simpler materials by ordinary chemical processing.
A molecule consists of two or more atoms. The process whereby the atoms stay together to form a molecule involves electron rearrangements. This process usually forms a molecule that acts much differently from the separate atoms of which it consists.
-
Question 7
Tell how atoms of metals differ from atoms of nonmetals in their electron arrangement. Why are metals good conductors?
Reveal answerAtoms of metals have only one, two, or three electrons in their outermost layer of electrons. These electrons are freely movable. Since conduction is the flow of electrons, the easy movability of these outer electrons is responsible for the fact that metals are good conductors.
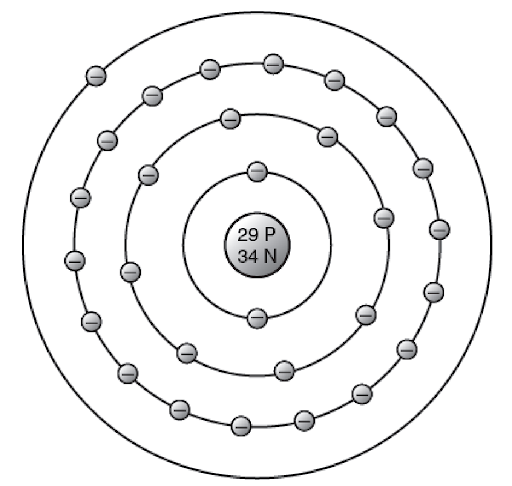
Metallic Element (Copper)
Nonmetallic elements have five, six, or seven outside electrons. These electrons are tightly held to the atoms and are not free to move. Thus, nonmetallic elements are not conductors.
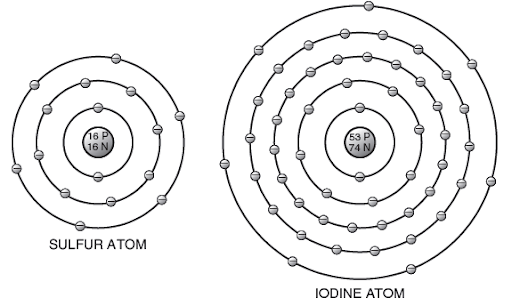
Nonmetallic Elements
Elements whose atoms have four outer electrons, such as carbon, silicon, and germanium, are often called semiconductors. How well these elements conduct depends on how the atoms are arranged with respect to each other, and the presence of impurities.
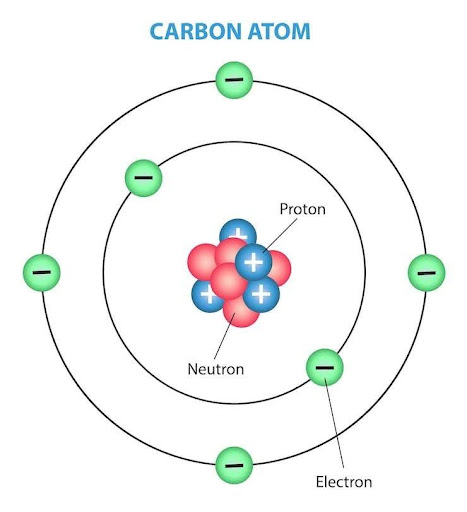
Semiconductor (Carbon)
-
Question 8
There is an element called gallium. Its atoms have 31 electrons. Referring to the picture of a copper atom, how would you expect the electrons of an atom of gallium to be arranged? Is gallium a metal?
Reveal answer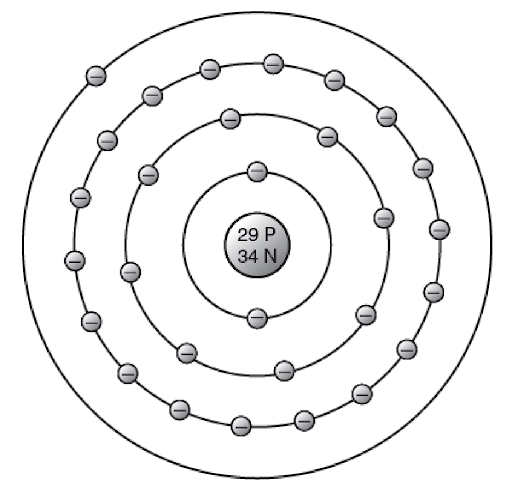
By referring to a picture of the copper atom, the probable arrangement of the 31 electrons of gallium can be listed. It is expected that the electrons are arranged as follows:
-
2 electrons in the innermost ring
-
8 electrons in the next ring
-
18 electrons in the next ring
-
3 electrons in the outer ring
This arrangement means that gallium is a metal.
-
-
Question 9
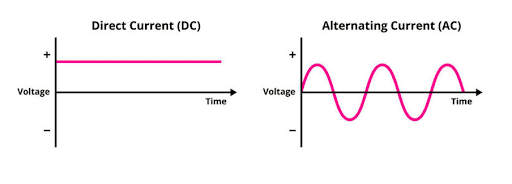
Explain the terms AC and DC. Tell how they differ from each other.
Reveal answerAC stands for Alternating Current, and DC stands for Direct Current.
AC-power sources have a continuously changing polarity. Consequently, the current in such circuitry alternates, back and forth, periodically. By contrast, the polarity of DC sources remains fixed and, therefore, the current always flows in the same direction (from negative to positive).